Introduction
Obesity is an emerging global health issue associated mainly with the development of numerous metabolic disorders, including dyslipidemia, type 2 diabetes, and cardiovascular disease [1-3]. A wealth of information illustrates that multiple factors such as genetics, individual demographic, and daily activity (sedentary lifestyle, lack of exercise, or poor eating habits) contribute to the induction and progression of obesity [4,5]. At the cellular and molecular levels, obesity is induced due to the abnormal expansion of white adipose tissue (WAT) under an energy surplus condition, which occurs mainly by an increase in the (pre)adipocyte size and number [6-8]. Excessive lipid accumulation in the preadipocyte differentiation results in hypertrophic adipocytes. Thus, any natural substance that represses excessive lipid accumulation in the preadipocyte differentiation process could be a potential obesity preventive and therapeutic reagent.
Preadipocyte differentiation, also called adipogenesis, is a complex process of adipocyte maturation from preadipocytes. The process is controlled by multiple transcription factors, which include peroxisome proliferator-activated receptors (PPARs), CCAAT/enhancer-binding proteins (C/EBPs), and signal transducer and activator of transcription (STAT) proteins [9-11]. It also has been illustrated that some lipogenic enzymes and lipid droplet (LD)-binding/stabilizing proteins, such as fatty acid synthase (FAS), acetyl-CoA carboxylase (ACC), and perilipin A, are necessary to promote fat synthesis (lipogenesis) and storage during preadipocyte differentiation and adipocyte maturation [12-14].
Salvianolic acid B (Sal B) is one of the significant bioactive hydrophilic compounds derived from the dry root and rhizome of Salvia miltiorrhiza [15,16]. Sal B has been widely utilized as a traditional medicine to treat cardio and cerebrovascular diseases. In recent years, some in vivo and in vitro studies have shown that Sal B has anti-obesity effects by suppressing the expression of PPAR-γ, C/EBP-α, and GATA binding protein-2/3 (GATA-2/3) [17-21]. Moreover, Sal B also found to have effects in lipid metabolism by reducing fasting blood glucose, total cholesterol, triglycerides (TGs), low-density lipoprotein cholesterol, and increasing high-density lipoprotein levels in mice [22]. However, other studies demonstrated that Sal B could promote adipocyte differentiation in 3T3-L1 cells by increasing PPAR-γ, PPAR-α, and C/EBP-α mRNA expression levels and improve fat graft survival [23,24].
So far, Sal B’s lipid-lowering effect and mode of action in adipocytes remain controversial and not fully understood. In this study, we therefore investigated and revealed the effect and different mechanism of Sal B in modulating lipid accumulation during the adipocyte differentiation of 3T3-L1 mouse preadipocytes. Our results demonstrate, for the first time, that Sal B at 100 µM vastly reduces lipid accumulation and TG content during 3T3-L1 preadipocyte differentiation, and Sal B's reductive effect is partly mediated not only through regulating the expression of C/EBP-α and PPAR-γ, but also STAT-3/5, FAS, ACC, and perilipin A.
Materials and methods
1. Materials
Sal B was purchased from Sigma Aldrich (St. Louis, MO, USA). Primary antibodies for anti-C/EBP-α, anti-phospho (p)-STAT-3, anti-STAT-3, anti-p-STAT-5, anti-STAT-5, anti-PPAR-γ were purchased from Santa Cruz Biotechnology (Delaware, CA, USA). The primary FAS antibody was obtained from BD Bioscience (San Jose, CA, USA). p-ACC and t-ACC were acquired from Cell Signaling Biotechnology Inc. (Danvers, MA, USA). Primary antibodies for perilipin A and β-actin were bought from Bio Vision (Milpitas, CA, USA) and Sigma (St. Louis, MO, USA), respectively.
2. Cell culture and differentiation of 3T3-L1 preadipocytes
3T3-L1 preadipocytes (ATCC, Manassas, VA, USA) were grown in the culture media containing Dulbecco’s Modified Eagles’ Medium (DMEM) (Welgene, Daegu, Korea) containing 10% heat-inactivated fetal calf serum (Gibco, Grand Island, NY, USA) and 1% penicillin/streptomycin (Welgene) at 37oC in a humidified atmosphere of 5% CO2. As shown in Fig. 1A, the differentiation of 3T3-L1 cells was initiated by replacing the culture media with new DMEM containing 10% fetal bovine serum (FBS) (Welgene), 0.5 mM IBMX (M) (Sigma), 0.5 µM dexamethasone (D) (Sigma), and 5 µg/ml insulin (I) (Sigma), abbreviated as MDI, either with or without Sal B. On day 2, the first differentiation medium was replaced with DMEM supplemented with 10% FBS and 5 µg/ml insulin either with or without Sal B at the indicated doses for additional 3 days. The cells were further fed with DMEM containing 10% FBS in the presence or absence of Sal B for other 3 days.
3. Oil red O staining
On day 8 of the post-differentiation induction, control or Sal B-treated 3T3-L1 cells were washed with phosphate-buffered saline (PBS) and fixed with 10% formaldehyde for 2 h. Cells were then washed with 60% isopropanol and dried. Oil red O working solution was added to the fixed cells for 1 h and washed with distilled water. Afterward, lipid droplets (LDs) accumulated in the conditioned cells were visualized under light microscopy (Nikon, Tokyo, Japan).
4. Cell count analysis
On day 8 of the post-differentiation induction, control or Sal B-treated 3T3-L1 cells were stained with trypan blue dye. Only cells with intact membranes can constructively exclude the dye, then dead cells with damaged membranes become stained and counted using a light microscope. The cell count assay was done in triplicates. Data are mean ± standard error (SE) of three independent experiments.
5. AdipoRed assay
On day 8 of the post-differentiation induction, TG content in control or Sal B-treated 3T3-L1 cells was analyze by AdipoRed Assay Reagent kit according to the company’s instructions (Lonza, Basel, Switzerland). After 10-min incubation, the plates were read in the Victor3 (PerkinElmer, Shelton, CT, USA), and fluorescence was measured with an excitation wavelength of 485 nm and an emission wavelength of 572 nm.
6. Preparation of whole-cell lysates
At treatments, 3T3-L1 cells were washed with PBS and lysed in a modified RIPA buffer (50 mM Tris-Cl [pH 7.4], 150 mM NaCl, 0.1% sodium dodecyl sulfate, 0.25% sodium deoxycholate, 1% Triton X-100, 1% Nonidet P-40, 1 mM EDTA, 1 mM EGTA, proteinase inhibitor cocktail [1x]). The whole-cell lysates were collected and centrifuged at 14,000 rpm for 15 min at 4°C. The supernatant was saved, and protein concentrations were determined with Pierce BCA Protein Assay Kit (Thermo Scientific, Rockford, IL, USA).
7. Western blot analysis
An aliquot of proteins (40 μg per lane) was separated by SDS-PAGE (10%) and then transferred to nitrocellulose membranes (Millipore). The membranes were washed with Tris-buffered saline (10 mM Tris, 150 mM NaCl) supplemented with 0.05% (v/v) Tween 20 (TBST) and blocked with blocking buffer (TBST containing 5% (w/v) non-fat dried milk). The membranes were incubated overnight with corresponding primary antibodies for C/EBP-α (1:1,000), PPAR-γ (1:1,000), p-STAT-3 (1:1,000), STAT-3 (1:1,000), p-STAT-5 (1:1,000), STAT-5 (1:1,000), FAS (1:1,000), p-ACC (1:1,000), t-ACC (1:1,000), perilipin A (1:1,000) or β-actin (1:10,000) at 4°C. The membranes were washed with TBST and then incubated with secondary antibodies coupled to horseradish peroxidase for 2 h. The membranes were then washed with TBST. ECL reagents was used to develop the image (Advansta, San Jose, CA, USA). Equal loading of the amounts of proteins per lane was verified by the β-actin antibody.
8. Quantitative real-time polymerase chain reaction analysis
Total RNA from control or Sal B-treated 3T3-L1 cells was isolated using RNAiso Plus (Takara, Kusatsu, Japan). Random hexadeoxynucleotide primer and reverse transcriptase were used for reverse transcribing an aliquot of total RNA (3 μg). The single-strand cDNA was amplified by polymerase chain reaction (PCR) with primers of C/EBP-α sense 5’-TTACAACAGGCCAGGTTTCC-3’; antisense 5’-CTCTGGGATGGATCGATTGT-3’; PPAR-γ sense 5’-GGTGAAACTCTGGGAGATTC-3’; antisense 5’-CAACCATTGGGTCAGCTCTC-3’; FAS sense 5’-TTGCTGGCACTACAGAATGC-3’; antisense 5’-AACAGCCTCAGAGCGACAAT-3’; ACC sense 5’-CAAGTGCTCAAGTTTGGCGC-3’; antisense 5’-CAAGAACCACCCCGAAGCTC-3’; Perilipin A sense 5’-CTTTCTCGACACACCATGGAAAC-3’; antisense 5’- CCACGTTATCCGTAACACCCTTCA-3’; 18S rRNA sense 5’-GGTGAAGGTCGGTGTGAACG-3’; antisense 5’-GGTAGGAACACGGAAGGCCA-3’. PCR reactions were performed in duplicate for each sample. The mRNA expression levels of each target gene were normalized to those of 18S ribosomal RNA (rRNA).
9. Statistical analysis
Cell count analysis was measured in triplicate and repeated three times. The results were expressed as mean ± SE. One-way analysis of variance was used to compare the difference significance. All significance testing was established on a p-value of < 0.05. The statistical software used in this study was the IBM SPPS Statistics 25 software (IBM, Armonk, NY, USA).
Results
1. Sal B inhibits lipid accumulation and reduces TG content in a concentration-dependent manner during 3T3-L1 preadipocyte differentiation
The experimental design for 3T3-L1 preadipocyte differentiation is illustrated in Fig. 1A. First of all, we investigated the effect of Sal B (Fig. 1B)at different doses (25, 50, and 100 μM) on lipid accumulation during the adipocyte differentiation of 3T3-L1 preadipocytes for 8 days, followed measurement of intracellular lipid droplet (LD) deposition by using Oil Red O staining. As shown in Fig. 1c (upper panel), compared with undifferentiated cells at D0, treatment with Sal B led to a concentration-dependent suppression of LD accumulation in 3T3-L1 cells of the differentiation on day 8. Sal B's LD-lowering effects in 3T3-L1 preadipocyte differentiation on D8 were further confirmed by phase-contrast image analysis (Fig. 1c, lower panel). We next probed whether Sal B could lower TG content in differentiating 3T3-L1 preadipocytes by using AdipoRed assay. As shown in Fig. 1D, treatment with Sal B at 25 or 50 μM did not influence TG content, but that with Sal B at 100 μM resulted in a significant reduction of TG content in 3T3-L1 cells of the differentiation on day 8.
2. Sal B at concentrations tested does not alter cell growth during 3T3-L1 preadipocyte differentiation
To see any cytotoxicity, we next tested whether Sal B at dosed applied alters cell growth (survival) in differentiating 3T3-L1 preadipocytes using a cell count analysis. As shown in Fig. 2, Sal B treatment at doses applied above had no effect on cell growth (survival) in 3T3-L1 cells on D8 of differentiation. Thus, due to the reductive maximal effects on LD accumulation and TG content with no cell growth inhibition, we chose the 100 μM of Sal B for further experiments.
3. Sal B at 100 μM down-regulates the protein and mRNA expression of C/EBP-α and PPAR-γ during 3T3-L1 preadipocyte differentiation
To unravel molecular mechanisms underlying Sal B’s LD-lowering effects herein, we investigated the effects of Sal B (100 μM) on the protein and mRNA expression levels of C/EBP-α and PPAR-γ, two adipogenic transcription factors in differentiating 3T3-L1 preadipocytes. Interestingly, as shown in Fig. 3A, Western blotting results demonstrated that treatment with Sal B led to a substantial decrease in the protein expression levels of C/EBP-α and PPAR-γ in 3T3-L1 cells on D5 and D8 of differentiation. However, the expression levels of β-actin, used as a protein loading and internal control to relatively compare those of target proteins herein (C/EBP-α and PPAR-γ), remained constant under these experimental conditions. As further shown in Fig. 3B, real-time qPCR results revealed the ability of Sal B to lower the transcripts of C/EBP-α and PPAR-γ in 3T3-L1 cells on D5 and D8 of differentiation in which the expression levels of 18S rRNA were used to normalize those of C/EBP-α and PPAR-γ.
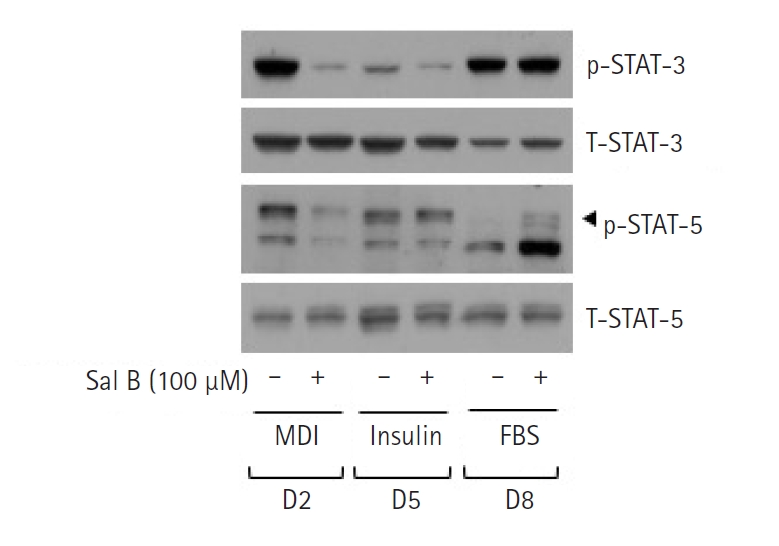
4. Sal B at 100 μM reduces the protein phosphorylation levels of STAT-3/5 during 3T3-L1 preadipocyte differentiation
We also determined whether Sal B (100 μM) affects the protein phosphorylation levels of STAT-3/5, other adipogenic transcription factors in differentiating 3T3-L1 preadipocytes. Strikingly, as shown in Fig. 4, treatment with Sal B caused a marked decrease in the protein phosphorylation of STAT-3/5 in 3T3-L1 cells on D2 of differentiation. Treatment with Sal B also had a mild suppressive effect on the protein phosphorylation of STAT-3/5 in 3T3-L1 cells on D5 of differentiation. Sal B treatment did not affect the protein phosphorylation of STAT-3, but it enhanced that of STAT-5 in 3T3-L1 cells on D8 of differentiation. Total expression levels of STAT-3/5 remained essentially unchanged under these experimental conditions.
5. Sal B at 100 μM lowers the protein expression and phosphorylation levels of FAS, ACC, and perilipin A during 3T3-L1 preadipocyte differentiation
We also sought to investigate whether Sal B (100 μM) alters the expression and phosphorylation levels of FAS, ACC, and perilipin A in 3T3-L1 cells on D2, D5, and D8 of differentiation. As shown in Fig. 5A, Sal B treatment resulted in sustained repression of the protein expression levels of FAS in 3T3-L1 cells on D2, D5, and D8 of differentiation. Sal B treatment also substantially declined ACC's protein phosphorylation and expression levels in differentiating 3T3-L1 preadipocytes on days tested herein. Treatment with Sal B caused potent inhibition of the protein expression levels of perilipin A in 3T3-L1 cells on D5 and D8 of differentiation. However, the expression levels of β-actin remained constant under these experimental conditions. Moreover, as shown in Fig. 5B, real-time qPCR results showed that Sal B treatment led to a time-dependent decrease in FAS, ACC, and perilipin A transcripts in 3T3-L1 cells on D5 and D8 of differentiation.
Discussion
Several lines of evidence illustrate that Sal B has lipid-modulating effects, but its anti-obesity effect and mechanism remain unclear and controversial. In this study, we investigated the effects of Sal B on lipid accumulation and TG content in 3T3-L1 preadipocyte differentiation. Here we demonstrate that Sal B at 100 µM has strong anti-adipogenic and anti-lipogenic effects on differentiating 3T3-L1 preadipocytes. Our results further suggest that these effects are mediated through control of the expression and phosphorylation levels of C/EBP-α, PPAR-γ, STAT-3/5, FAS, and perilipin A.
Previous in vitro and in vivo studies have shown that Sal B has both anti-adipogenic and pro-adipogenic effects [25-26]. In agreement with the former, the present study has demonstrated Sal B's lipid-lowering effects on differentiating 3T3-L1 preadipocytes, as assessed by the ability of this natural substance to inhibit LD accumulation and reduce TG content.
A notable finding of the present study is Sal B's regulation of the expression and phosphorylation (activation) of multiple adipogenesis-related transcription factors, including C/EBP-α, PPAR-γ, and STAT-3/5. It is well-documented that C/EBP-α and PPAR-γ are highly expressed at the initial stage of preadipocyte differentiation in response to C/EBP-β and C/EBP-δ activation, and the up-regulated C/EBP-α and PPAR-γ are pivotal in lipid accumulation at middle and late stages during preadipocyte differentiation [27-29]. There is also a wealth of information to support that STAT-3/5, two members of the STATs family, are up-regulated and hyperphosphorylated (hyperactivated) during preadipocyte differentiation, and the hyperactive and abundant expression of STAT-3/5 play crucial roles in adipogenesis at the early stage of preadipocyte differentiation [30-32]. Considering the present findings that differentiating 3T3-L1 cells treated with Sal B express less expression and phosphorylation of C/EBP-α, PPAR-γ, and STAT-3/5, it is suggested that Sal B exerts its lipid-lowering effects herein through the down-regulation of these transcription factors. Moreover, the present study illustrates that Sal B down-regulation of C/EBP-α and PPAR-γ in differentiating 3T3-L1 cells is at the levels of their transcriptional repression. In contrast, Sal B down-regulation of STAT-3/5 phosphorylation in differentiating 3T3-L1 cells is not through a change of de novo protein synthesis of STAT-3/5 but through the dephosphorylation of preexisted STAT-3/5 proteins, further pointing out the ability of Sal B to regulate post-translational modification of these transcriptional factors.
Another interesting finding in this work is Sal B's regulation of the expression of FAS, ACC, and perilipin A. As aforementioned, FAS and ACC are lipogenic enzymes involved in synthesizing fatty acids, and their expressions and activities are elevated during preadipocyte differentiation [33,34]. Perilipin A is the most abundant protein that binds to and stabilizes intracellular LD, which plays a vital role in fat accumulation or storage in differentiating preadipocytes and mature adipocytes [35,36]. Until now, Sal B regulation of FAS, ACC, and perilipin A expressions in preadipocyte differentiation process is not reported yet. Of importance, we herein demonstrated that Sal B could strongly repress the expression of FAS, ACC, and perilipin A at both protein and mRNA levels in differentiating 3T3-L1 cells at the middle and late stages of differentiation. These results thus indicate that the reduced expressions of FAS, ACC, and perilipin A further contribute to Sal B’s lipid-lowering effect on differentiating 3T3-L1 cells.
Conclusion
It is the first report that Sal B has strong anti-adipogenic effects on differentiating 3T3-L1 cells, partly mediated through the reduced expression and activity of C/EBP-α, PPAR-γ, STAT-3/5, FAS, and perilipin A. This work suggests Sal B as a potential obesity preventive and therapeutic agent that inhibits lipid accumulation or storage in differentiating preadipocytes.